Analgesic cleansing composition
US Patent 9,364,402
Application No 14/587,950
Granted 14 June 2016
Assignee The Dial Corporation
Hand sanitiser compositions have antibacterial properties and cleanse the hands without the need for direct contact with water. Topical analgesic compositions provide relief from muscle and ligament soreness and existing ones are applied as leave-on creams or gels to provide extended contact. The patent describes analgesic cleansing compositions, which may be either wash-off or leave-on, and may optionally include antibacterial agents and a cosmetically suitable carrier.
The patent names many possible analgesics both natural and synthetic, but methyl salicylate is the one preferred. It names virtually every class of surfactant without showing any preference and example compositions of wash-off compositions show 4-10% anionic surfactant content in combination with non-ionic and zwitterionic surfactants, a deposition aid and up to 10% methyl lactate. Leave-on compositions have a maximum 1.5% anionic surfactant, 0.5-10% methyl lactate and a deposition aid.
Examples of deposition aids are cationic polymers; polyquaternium-7, polyquaternium-10 and polyquaternium-11 are named.
Leave-on cleansing compositions are provided in an aqueous-alcoholic carrier containing at least 50% ethanol or isopropanol. Both types of cleansing composition may also contain a polymeric thickening aid, an antibacterial agent, fragrance, humectants, an exfoliant, a conditioning agent, plant extracts and other additives to enhance product aesthetics, shelf life and consumer appeal.
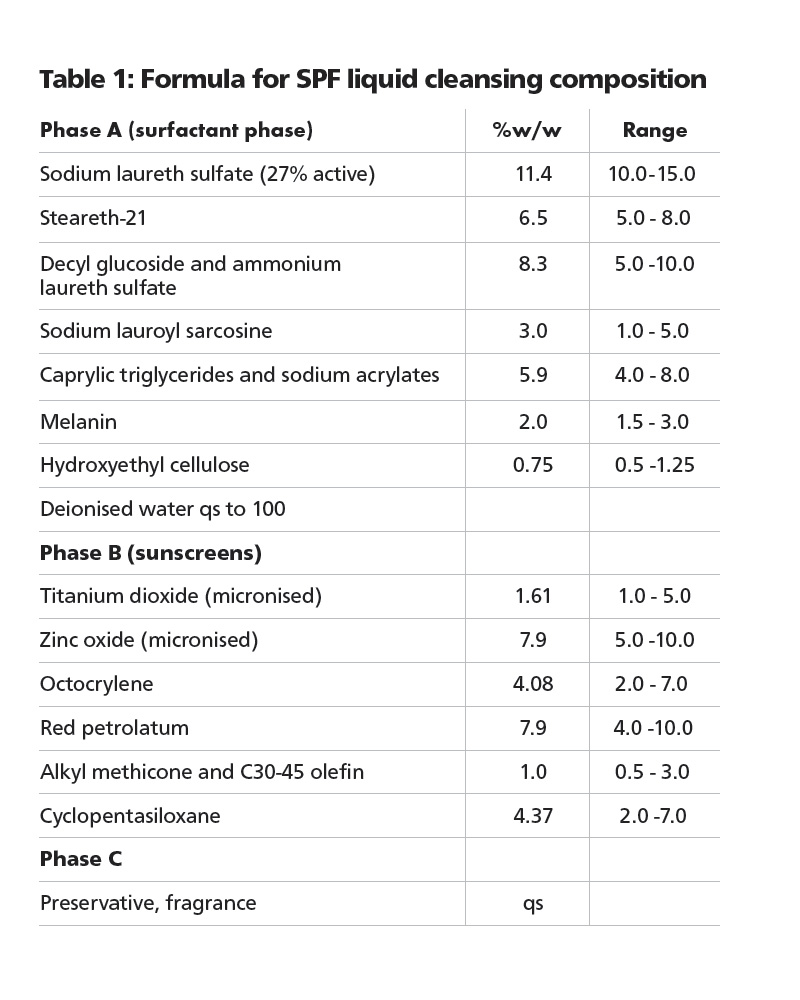
SPF liquid cleansing compositions and methods of use
US Patent 9,351,913
Application No 14/530,787
Granted 31 May 2016
Assignee Cockerell
Dermatology Development The patent describes a facial or body wash composition that, after rinsing, provides a sun protection factor of at least six.
The preferred surfactant system is a composition of ammonium laureth sulfate in combination decyl glucoside. Additional surfactants are sodium laureth sulfate, steareth-21 and sodium lauroyl sarcosine.
The applicants claim that red petrolatum is an essential element of the composition and additionally, the composition contains an organic sunscreen such as octocrylene and micronised zinc oxide titanium dioxide, surface treated to render them hydrophobic. Other essential ingredients are an alkyl silicone, a volatile cyclic silicone such as cyclopentasiloxane and melanin or a melanin precursor.
Optionally, it may also contain iron oxide pigments. The metal oxides and pigments block ultraviolet radiation in the wavelength range of from about 290nm to about 400nm.
Deposition of the sunscreen is assisted by the inclusion of sodium acrylate in capric triglyceride – available as Luvigel EM from BASF – and hydroxyethyl cellulose for viscosity control. The composition may also include preservatives, perfume etc and is preferably supplied in aerosol format.
After mixing, the composition serves as a concentrate and is combined with propellant at a ratio of 96% concentrate to 4% propellant in an aerosol container. Upon application to the skin, the product is a shiny, flowable, tinted product which, within seconds, increases in volume and appears as a mousse. After cleansing and rinsing according to standard test protocols an in vivo test showed UVB protection of SPF7.9.



