Hyaluronic acid gel and manufacturing method thereof
US Patent 9,855,206
Application No 14/436,463
Granted 2 January 2018
Assignee Cosmed Pharmaceutical Co.
Hyaluronic acid is contained in the human body and has excellent biocompatibility and moisture retaining action.
It is a linear polymeric polysaccharide prepared by isolation and extraction from the crests and umbilical cords of chickens, or by fermentation using microorganisms such as Streptococcus.
Hyaluronic acid gel is usually prepared by crosslinking hyaluronic acid through chemical modification. However, such gel is poorly water soluble, and it is unsuitable for providing moisture and tension to the skin.
The patent describes a hyaluronic acid gel suitable for cosmetic and medical use comprising hyaluronic acid, a polycarboxylic acid and a polyhydric alcohol.
It also contains at least one or more compounds selected from a group consisting of hydroxypropylcellulose, polyvinylpyrrolidone, polyvinyl alcohol, carboxymethylcellulose, polyacrylic acid and polyethylene glycol.
It is claimed that, by exploiting the properties of hyaluronic acid, the gel has flexibility, elasticity and tensile strength, and will adhere tightly to the skin to moisturise it.
Furthermore, if the gel is applied to skin and the skin is then massaged with water, the gel will rapidly dissolve, and the hyaluronic acid and other ingredients will be absorbed by the skin.
Additionally, when the gel is applied to skin, a large amount of the polyhydric alcohol contained in the gel is hydrated with water to generate heat, and it gives a mild warming sensation.
The polycarboxylic acid is selected from a group consisting of citric acid, tartaric acid and lactic acid, and the preferred polyhydric alcohol is glycerin.
The gel may also contain whitening, anti-wrinkle, anti-itching and anti-inflammatory ingredients, blood circulation promoting components, antimicrobials and antioxidants, and various vitamins and their derivatives, plus pigments, fragrances and other cosmetically beneficial materials.
Aqueous solutions of each component are mixed together to form a homogenous solution, which is then poured into moulds and the moisture content evaporated to form a suitable sheet shape such as a whole-face sheet or an eye sheet, the thickness of which is preferably 30μm-1mm.
The patent describes various weight ratios of the components and how gel sheets were tested for flexibility, tensile strength, adherence to skin and moisturising effects.
Formation of hydrated nanocellulose sheets with or without a binder for the use as a dermatological treatment
US Patent 9,816,230
Application No 14/986,578
Granted 14 November 2017
Assignee Innovatech Engineering
Hydrogels are often used for dermatological masks because of the large amount of water they can hold. Unfortunately, these masks have a low degree of conformability to the skin and are not porous.
Described is a method for manufacturing a hydrated non-woven nanocellulose sheet having dermatologically active ingredients.
The sheet is formed through a high pressure or vacuum filtration process from a dilute suspension of the nanocellulose.
The suspension may also contain dermatologically active ingredients and binding agents that improve the strength of the sheet.
These may be crosslinked after the formation of the sheet by applying chemical agents. Manufacture involves providing purified nanocellulose into a 'first suspension' to a mass concentration of up to 10g-per-litre in water, alcohol or oil.
The suspension is filtered with positive pressure or vacuum to form a sheet, which is dipped into a solution of crosslinking agents such as calcium citrate, lactate, chloride or stearate.
It is then dipped into an ingredient slurry to absorb any actives required. The sheet is then cut into a form, such as a facial mask, neck wrap or under-eye masks and sealed into an airtight package.
Active ingredient combinations of magnolia bark extract and hyaluronic acid and the cosmetic and/or dermatological use thereof
US Patent 9,402,801
Application No 14/411,688
Granted 2 August 2016
Assignee Beiersdorf
The patent relates to active ingredient combinations of magnolia bark extract and hyaluronic acid preparations, and their use for the treatment or prophylaxis of cellulite and the appearance of skin ageing.
The preparations further comprise anise fruit extract (Pimpinella anisum fruit extract) obtained by enzymatic hydrolysis of anise fruits solubilised in water.
Anise fruit extract is rich in inorganic minerals such as sodium and magnesium ions, but particularly in potassium ions.
Claimed is a topical cosmetic or dermatological preparation comprising from 0.05-5% of magnolia bark extract, from 0.05-5% of hyaluronic acid and 0.1-15% of anise fruit extract.
The preparation is a cream, lotion or cosmetic milk in emulsion form, and it is claimed that when applied to areas of the skin with cellulite the skin is tightened and firmed.
Skin moisture is also increased and lipid synthesis stimulated. The oil phase is described at some length and, of all the materials mentioned, mixtures of cyclomethicone and isotridecyl isononanoate or of cyclomethicone and 2-ethylhexyl isostearate were cited as being particularly advantageous.
Other constituents added to improve aesthetics and shelf life include consistency regulators, fillers, perfumes, dyes, emulsifiers, additional active ingredients such as vitamins or proteins, light protection agents, stabilisers, insect repellents, alcohol, water, salts and antimicrobial, proteolytic or keratolytic substances.
Depilatory formulations and methods of using same
US Patent 9,795,558
Application No 14/418,948
Granted 24 October 2017
Assignee Reckitt & Colman
The applicants claim that depilatory formulations require a strong alkaline pH to achieve chemical cleavage of the keratin bond in the hair.
Formulations must be applied to a user's skin and left for a period of up to ten minutes prior to removal.
The high alkaline pH of such formulations can cause significant irritation to users' skins.
The patent describes a formulation suitable for utilisation as a pre-use depilatory ancillary, comprising a hydrophobic film-forming polymer and a suitable solvent.
The hydrophobic film-forming polymer is polyamide-3, the solvent is propylene glycol and the ratio of solvent to hydrophobic film-forming polymer is between 5:1 and 2:1.
The ratio of solvent to film-forming polymer is such that the formulation dries on the skin and forms a protective layer.
The pre-use depilatory formulation further comprises one or more excipients selected from the group consisting of skin feel enhancers, humectants, emollients, anti-irritation compounds and cosmetic actives.
The preparation is preferably formulated as an emulsion comprising between 1-10% silicone wax, between 1-10% of a mixture of mineral oil, silicone and emollient esters, plus a surfactant and a polymer.
The patent also claims depilatory combinations that comprise a pre-use formulation and a depilatory formulation.
The depilatory preparation contains a compound capable of degrading keratin and many such materials are listed.
This is present at preferably 10-15% in aqueous solution and the pH is adjusted by the addition of a suitable hydroxide so that the pH is preferably 9-12.5.
A non-ionic surfactant and urea to accelerate the reaction plus suitable additives to improve the aesthetics and stability of the preparation are included.
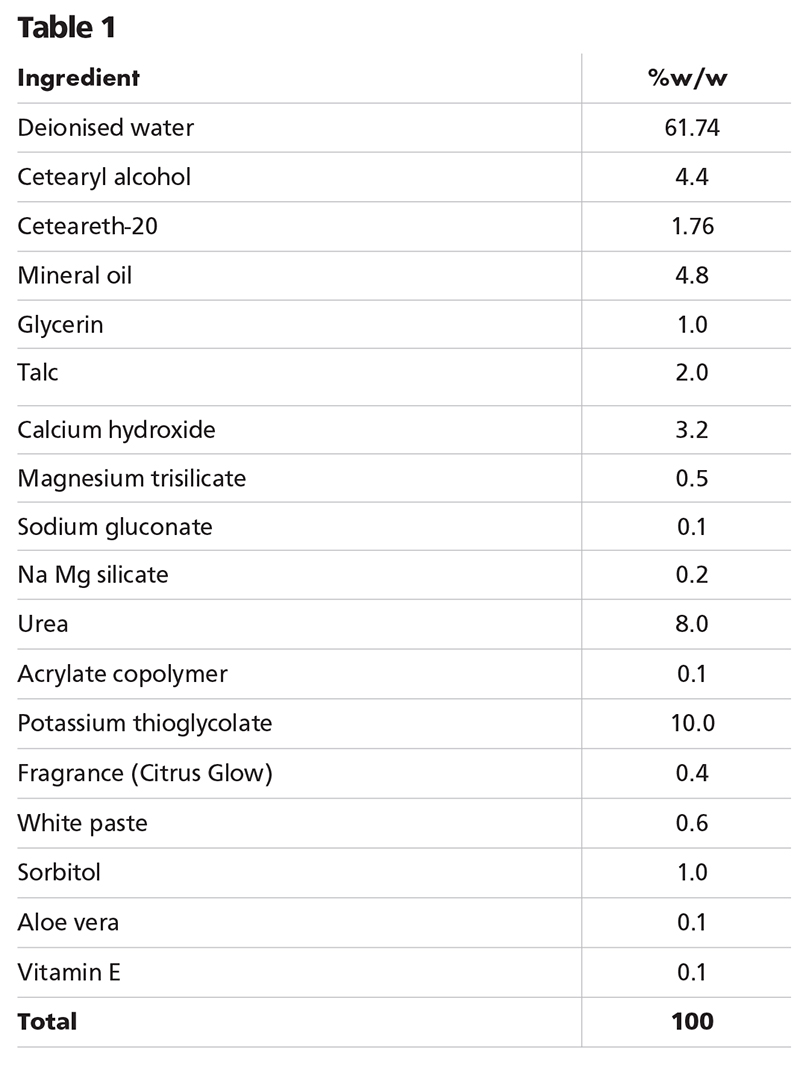
An example for the depilatory is shown in table 1.