With the ever-growing popularity of sunscreens, it is important as a formulator to use the most effective and efficient strategies to maximise the protection potential of each UV filter within a formulation.
Studies have shown that exploiting the synergistic effects of a combination of organic and inorganic UV filters can greatly benefit the overall formulation by allowing for higher solids loading, less interaction with organic UV filters and lower costs per unit of SPF.
The synergy benefits of using both types of UV filters are well documented and are based upon the different mechanisms of protection from each type of UV filter working differently to each other but also working well together.
This has previously been demonstrated in a study using the Monte Carlo calculation which used typical optical parameters of the skin to characterise the synergy effect when organic and inorganic UV filters are used together.
This proved that the synergistic performance is caused by light scattering from inorganic UV filters in combination with organic UV filters.1
As awareness of the harmful effects of UV light increases, the need for effective sun protection products becomes a necessity. At present in the UK alone there are approximately 16,200 new melanoma skin cancer cases every year (2015-2017) meaning the need for more sophisticated sun protection products is higher than ever.2
As awareness increases around the importance of SPF and UVA ratings, this means that sun protection products need to follow current trends, meet strict guidelines in terms of high SPF, UVA protection and meet sensorial requirements.
For the formulator, lots of factors must be taken into consideration. They not only face strict regulations to abide by which are different in each region, but the products must pass strict and expensive in vivo SPF and UVAPF tests whilst also taking into account the ethical and environmental implications of each product, all whilst keeping the sensory and the cost of the product desirable.
This is where taking advantage of the synergistic effects of using a combination of organic and inorganic UV filters can help within a formulation.3
Inorganic UV filters are often referred to as physical UV filters due to their mode of skin protection. The most used inorganic UV filters are Titanium Dioxide (TiO2) and Zinc Oxide (ZnO) which are growing in popularity due to the known mildness and lack of skin penetration and other factors such as natural mineral origin and reef-safe claims.
Formulators often combine TiO2 and ZnO due to the different absorbance curves and the good broadband UV protection when used together.4 TiO2 mostly protects within the UVB region with limited protection against the UVA region, however this is dependent on the particle size.
In contrast, ZnO has an absorbance range within both the UVB and UVA regions meaning they are usually combined to reach broad spectrum protection.5
Organic UV filters are commonly referred to as chemical UV filters due to their mode of skin protection which is different to that of inorganic UV filters.
Organic UV filters are usually aromatic compounds with a carbonyl group and examples of commonly used types are cinnamates, salicylates and benzophenones. In contrast to inorganic UV filters, usually a cocktail of organic UV filters is necessary within a formulation to achieve broad spectrum protection as they typically have narrow absorbance ranges.5
Experimental results
In vitro SPF evaluation
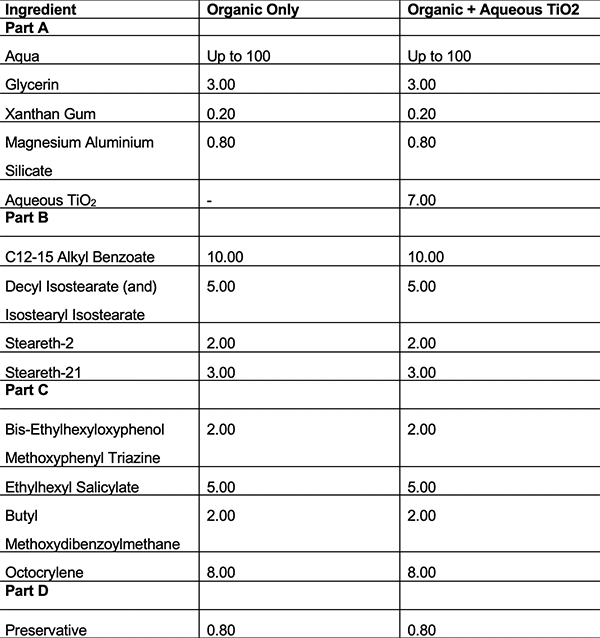
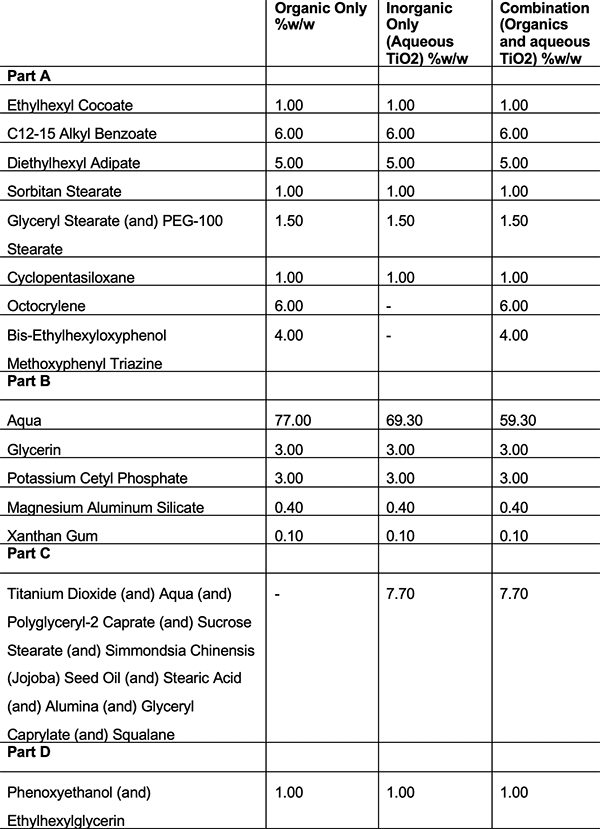
To explore this synergistic effect, six oil in water emulsifiers were formulated using two different UV filter systems. The first filter system being organic UV filters only and then the second filter system being with the same organic UV filters but with the addition of an aqueous TiO2 dispersion.
Using this formulation strategy meant it was possible to populate both phases of the formulation as the organic UV filters are oil soluble and present in the internal oil phase whereas the aqueous TiO2 dispersion is present in the external phase.
These formulations can be seen below in tables 1-6. Once formulated, these formulations underwent in vitro SPF testing to assess the differences in SPF between the UV filter systems and the in silico predicted SPF values.
Table 1: Formulations using emulsifier A
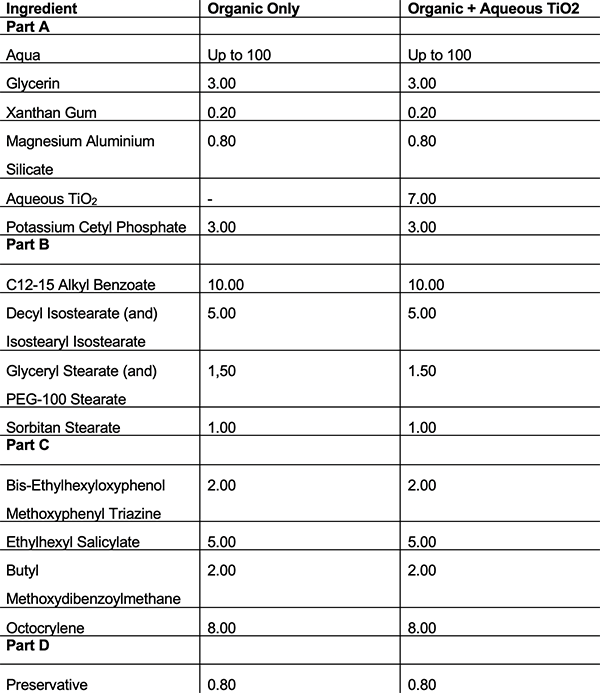
Table 2: Formulations using emulsifier B
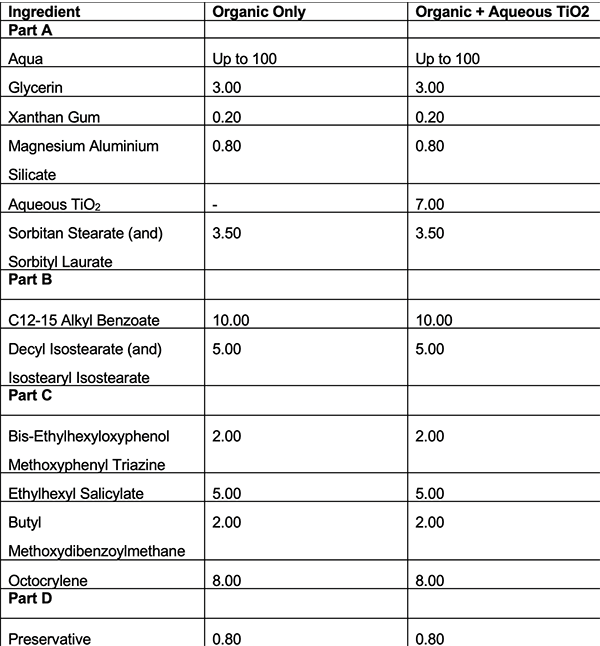
Table 3: Formulations using emulsifier C
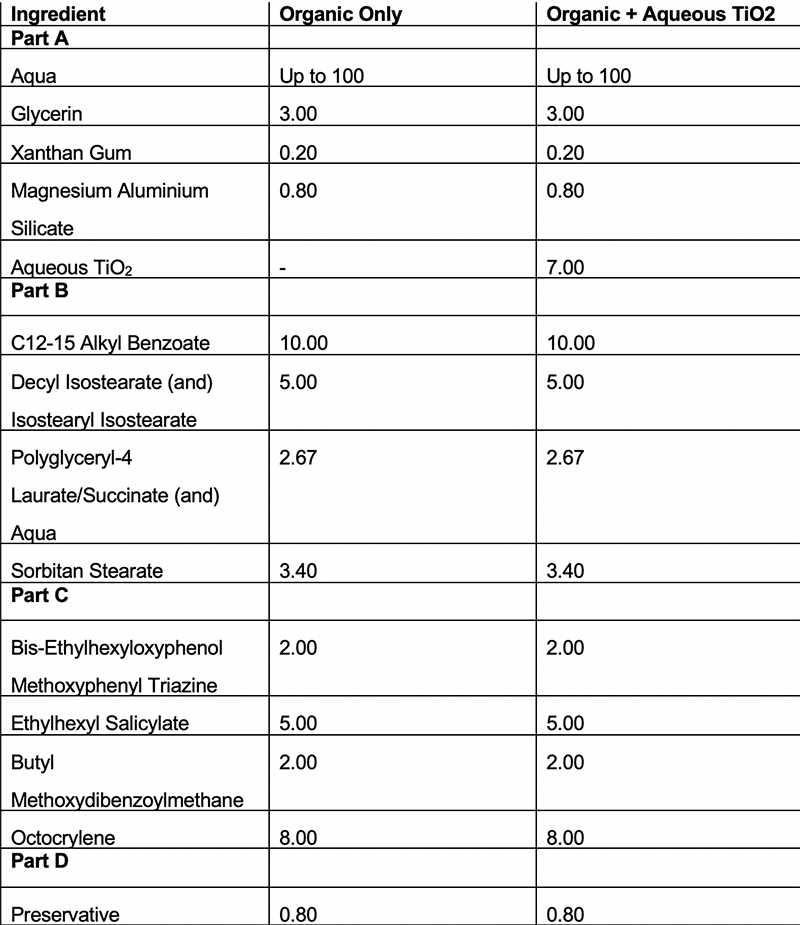
Table 4: Formulations using emulsifier D
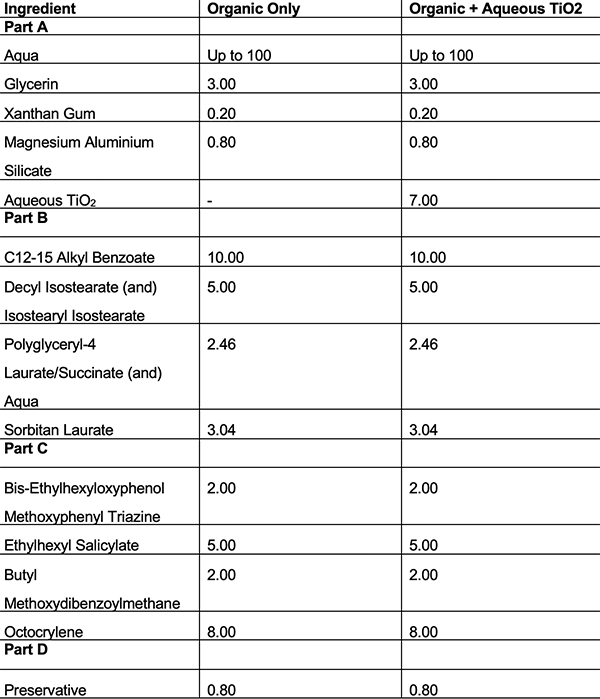
Table 5: Formulations using emulsifier E
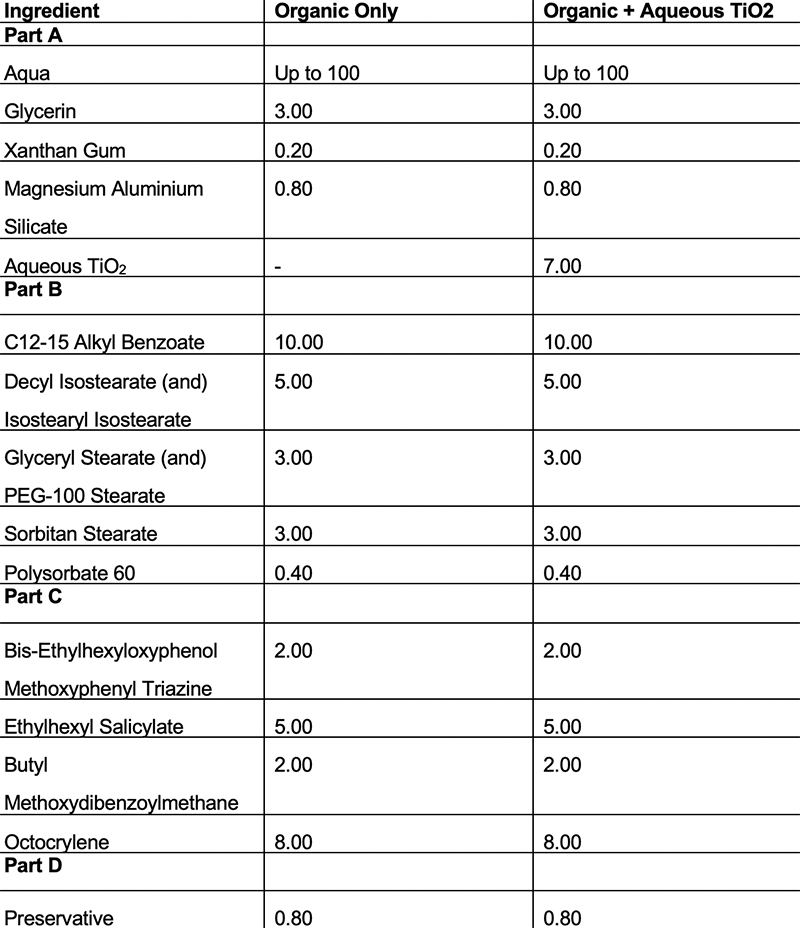
Table 6: Formulations using emulsifier F
For the twelve formulations with the six different emulsifier systems and two different UV filter systems above, the in vitro SPF was measured and compared to the expected in silico SPF values.
To measure the in vitro SPF, automated spreading was carried out using the HD-Spreadmaster (HelioScreen) onto two types of PMMA plates (HD6 and SB6) at 25C regulated using the HD-Thermaster (HelioScreen) and then measured using the UV2000S Labsphere.
As you can see in table 7 below the in vitro SPF values were consistently much higher in the formulations that used the combination of both organic UV filters and an aqueous TiO2 dispersion.
Table 7: SPF in-vitro for all six formulations (different emulsifiers)
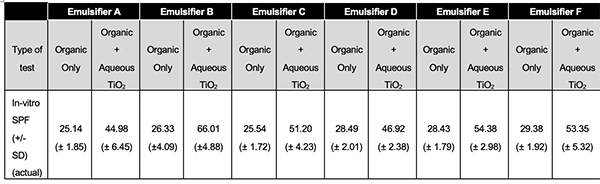
Figure 1 also shows the in vitro results of the twelve formulations with and without aqueous TiO2 dispersion along with the expected theoretical values.
As can be seen in figure 1 the in vitro results for all the systems with aqueous TiO2 has much higher in vitro SPF results than were predicted and thus could be representing the synergistic effect between the two types of UV filters.
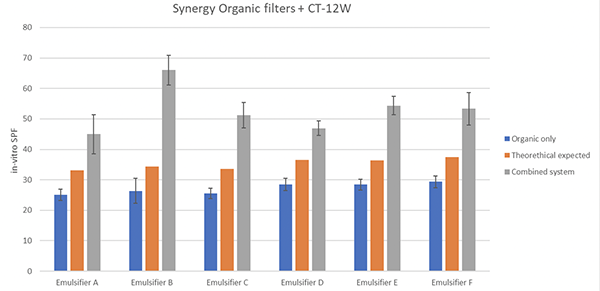
Figure 1. In-vitro SPF of formulations with different O/W emulsifiers and two UV filter systems (organic UV filters only and organic UV filters with aqueous TiO2 dispersion)
In vivo SPF and in vitro UVAPF evaluation
Another study was conducted whereby three formulations were made and sent for in vivo SPF and in vitro UVAPF testing which can be seen in table 8 below.
The three formulations were the same besides the UV filter combinations. One was made with just organic UV filters, the second with inorganic UV filters only and the third contained a combination of organic and inorganic UV filters.
Once made, these formulations were sent off for external in vivo SPF and internal in vitro UVAPF testing and the results can be seen below in figure 2.
Table 8: Formulations made for in vivo SPF analysis
As you can see in figure 2, there is a clear increase of in vivo SPF and in vitro UVAPF when organic and inorganic UV filters are combined compared to when they are used separately which again may be indicative of the synergy effect between both types of UV filters.

Figure 1. In-vitro SPF of formulations with different O/W emulsifiers and two UV filter systems (organic UV filters only and organic UV filters with aqueous TiO2 dispersion)
Discussion
As seen in the two sets of results above, our studies both in vitro and in vivo have found that when a combination of aqueous TiO2 dispersion is added alongside organic UV filters there is a significant increase in SPF and UVAPF units when tested compared to the same system that did not use a combination of UV filters.
It is thought that the reasoning behind this is down to the synergy effect and possibly the presence of UV filters in both phases of an emulsion resulting in an SPF boost since they are fully present and distributed in both phases.
Other referenced studies have used the Monte Carlo calculation to prove this theory by using typical optical parameters of the skin to characterise the synergy effect when organic and inorganic UV filters are used together.
They proved that the synergistic effect is caused by light scattering from inorganic UV filters in combination with organic UV filters. This theory is demonstrated below in figure 3.
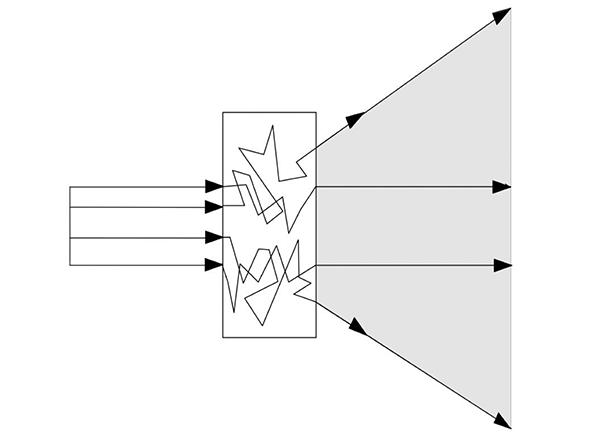
Figure 3. Increase in the optical pathway of photons in an absorbing medium on account of scattering
As discussed, aqueous TiO2 dispersion is recommended due to the lack of interaction with organic UV filters. There are many variations of aqueous TiO2 dispersions which have smaller or larger particle sizes which will ultimately affect the formulation in terms of the transparency on the skin and the range of UVA and UVB protection.
The benefits of using these aqueous dispersions is that they give you great synergy along with formulation flexibility.
Whilst formulating it is important to keep in mind that some combinations of organic and inorganic UV filters are not desirable such as the combination of Butyl Methoxydibenzoylmethane (BMDM) with TiO2.
This is because BMDM is known to form yellow coloured complexes when used alongside metal species ie TiO2. The yellow complexation does not actually reduce the UV protection of the filters, it is however undesirable from an aesthetics point of view to have a yellow coloured sunscreen.
However, if these two UV filters must be used together then there are things that can help. A good method would be to use a chelating agent in the formulation to help avoid complexation.
It is also recommended to use an organic compatible TiO2 such as grades with dense Silica coatings. This is because Silica is selected as having the lowest potential to interact with BMDM.7
Also, using an aqueous TiO2 dispersion means that there is no interaction with organic UV filters, which allows more optimisation of the oil phase meaning you can really take advantage of the formulation synergy.
It should however be considered and noted that the combination of BMDM and TiO2 in the USA under the current FDA’s proposed rules for sunscreen products is not permitted.6
An internal study demonstrated that the yellowing seen when TiO2 is used with BMDM is far less when aqueous TiO2 dispersion is used compared to the oil based TiO2 dispersion of the same particle size.
This test was performed using a colorimeter after one day, one week and once month. See graph below which shows the yellowing results when assessed. (The higher the b-value the greater the extent of the yellowing).
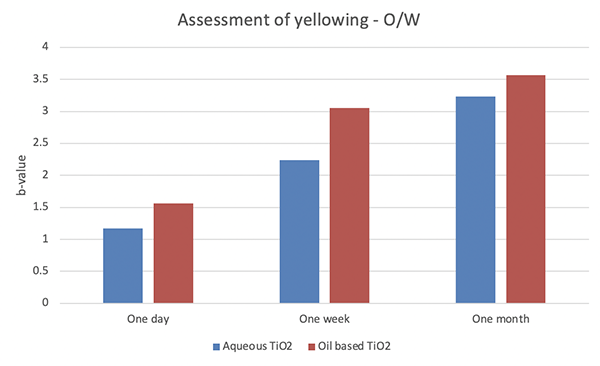
Figure 4. TiO2 dispersions and BMDM in an O/W formulation over time
As you can see in figure 3, there is a clear reduction in yellowing of a formulation when an aqueous TiO2 dispersion is used alongside BMDM compared to the oil based TiO2 dispersion. Again, this is thought to be down to the filters working well together as they are in separate phases of the emulsion.
Conclusion
The theory behind synergy is that when organic and inorganic UV filters are combined these UV filters provide two different mechanisms of protection rather than one (if used alone).
With an inorganic physical UV filter such as TiO2, the mechanism of protection is generated through three mechanisms which are reflection, scattering and absorption of the UV light.5
These inorganic filters have many advantages such as broad-spectrum protection, photostability and known mildness. With an organic UV filter, the mechanism of protection is through a chemical reaction that occurs which is initiated when UV light hits the UV filter.
When this happens there are conformational molecular changes that happen which is then released as both heat energy and radiation of a high wavelength.5
Organic UV filters may have some disadvantages such as a narrower spectrum of protection along with lower photostability which is why it can be beneficial to combine with inorganic UV filters.
However, when both UV filters are used together both mechanisms of protection are initiated leading to a synergy effect due to the physical protection mechanisms of the inorganic UV filters along with the absorption mechanisms of the organic UV filters, thus leading to more UV radiation being absorbed and reflected than if the products were used alone1.
In summary, it is useful to use a combination of organic and inorganic UV filters as TiO2 provides a broad-spectrum base in which to build upon. Organic UV filters are then used to build SPF and meet various regional requirements for UVA.
The synergistic effect between inorganic and organic UV filters is beneficial in that it helps to achieve higher SPF and UVA but also gives a lower cost per unit of active. This also means that this can be a cost-effective method of formulating high SPF sunscreens.
In addition to this, it is beneficial to use the synergy benefits of a combination of organic and inorganic UV filters as this allows for a higher solids loading within a formulation if the two types of UV filters are used in separate phases.
There are numerous methods of incorporating a combination of both types of UV filters into a formulation. However, the most effective method of incorporation is to use an aqueous dispersion of TiO2 alongside oil soluble organic UV filters.
References
1 .Lademann, J., Schanzer, S., Jacobi, U., Schaefer, H., Pflücker, F., Driller, H., Beck, J., Meinke, M., Roggan, A. and Sterry, W., 2005. Synergy effects between organic and inorganic UV filters in sunscreens. Journal of Biomedical Optics, 10(1), p.014008
2. Cancer Research UK. 2020. Melanoma Skin Cancer Statistics. [online] Available at:
3. Lim, H. and Draelos, Z., 2009. Clinical Guide to Sunscreens and Photoprotection. Basic and Clinical Dermatology, Volume 43. Informa Healthcare
4. Schneider, S. and Lim, H., 2018. A review of inorganic UV filters zinc oxide and titanium dioxide. Photodermatology, Photoimmunology & Photomedicine, 35(6), pp.442-446
5. Manaia, E., Kaminski, R., Correa, M., Chiavacci, L., 2013. Inorganic UV filters. Brazilian journal of pharmaceutical science, 49(2), pp. 201-209
6. Electronic Code of Federal Regulations (eCFR). 2020. Electronic Code of Federal Regulations (Ecfr). [online] Available at:
7. Egerton, T., Everall, N., Mattinson, J., Kessell, L. and Tooley, I., 2008. Interaction of TiO2 nano-particles with organic UV absorbers. Journal of Photochemistry and Photobiology A: Chemistry, 193(1), pp.10-17.

