British watchdog the Advertising Standards Authority (ASA) has banned a string of skin care adverts claiming to target acne.
Clearasil, Dermalogica, Feelunique and Johnson & Johnson (J&J) were all ordered to remove online campaigns that made ‘medicinal claims’.
A web page on Dermalogica’s website, promoting its MediBac clearing range, said the products are designed to “treat, clear and prevent adult acne” and “provide around-the-clock, maximum control of the main factors that contribute to acne”.
Dermalogica proposed to replace “acne” with the terms “breakout” or “blemish”.
However, the ASA believed the amendments would not be enough. It argued that acne is a medical condition and beauty product adverts that stated, or implied, a product could prevent or treat acne were medicinal claims.
The ASA said: “'Spots’ would most commonly be associated in public perception with acne, and therefore claims to prevent or treat them would also likely be seen as medicinal.”
Beauty online retailer Feelunique included a category page for a range of skin care brands “for acne and problem skin”.
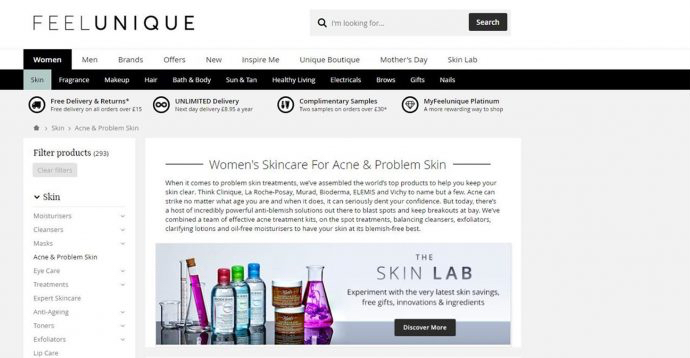
Feelunique's original claims
The UK company boasted to have “combined a team of effective acne treatment kits, on the spot treatments, balancing cleansers, exfoliators, clarifying lotions and oil-free moisturisers to have your skin at its blemish-free best.”
Feelunique argued that “acne” is a term broadly used by consumers and it was unlikely shoppers thought of the skin concern in the same way as medical conditions, such as "diabetes or appendicitis".
- Puig wins ASA ruling after Paco Rabanne fragrance ad receives 120 complaints
- Proactiv ad banned for suggesting children with acne will be bullied
- Made in Chelsea star breaches advertising rules with Vanity Planet Instagram story
Meanwhile, Reckitt Benckiser UK-owned Clearasil marketed its Rapid Action Treatment as “clinically proven to visibly reduce spot size and redness in as fast as four hours".
Similarly, J&J’s Clean and Clear claimed its Advantage Spot Control Treatment Gel contained a “unique spot-fighting formula [that] will quickly remove excess oil, helps fight problem-causing bacteria and purify skin”.
The ASA said all of the adverts must not appear in their current form, as the brands could not make medicinal claims for a primary beauty product.
