It is well-known that topical exposure to atmospheric pollutants (ie exhaust gases, heavy metals, free radicals and the sun’s rays) activates cutaneous stress and inflammatory response, which leads to reactive oxygen species (ROS) formation and decreases the antioxidant skin defence[1].
This represents one of the major concerns for the cosmetics industry and formulators. In this context, green algae, like spirulina (Arthrospira platensis) and those of the genus Chlorella provide a great variety of bioactive compounds of nutraceutical, pharmaceutical and cosmetic interest[2].
Belonging to the cyanobacteria taxon, Klamath algae (AFA, Aphanizomenon flos-aquae) is a water-based unicellular blue-green algae, which grows in the homonymous lake of volcanic origin (Upper Klamath Lake, Oregon, US).
Due to the extraordinary richness of its phytocomplex, AFA is nowadays of particular interest for the nutritional supplement industry, but until now, its cosmetic potentiality has not yet been developed.
Its most interesting asset is C-Phycocyanin, a well-known chromophore complex that confers the product a strong protective capacity against exogenous and endogenous oxidative damage of genomic and mitochondrial DNA[3].
AFA contains a wide range of trace elements (Fe, Zn, Se and Mg), a broad spectrum of vitamins (mainly of groups B, K and C), essential amino acids, PUFA Ω-3 (mainly α-linolenic) and the cellular growth factor GFC[4], which justify its use in numerous immunostimulant and anti-inflammatory nutraceutical products[5].
Figure 1:

What is AFA-GS and how does it work?
AFA also synthesises glyceryl glucoside, a glycerin derivative with remarkable skin moisturising properties. In order to evaluate the topical anti-pollution, anti-radical, antioxidant, sun’s rays-protection and anti-inflammatory potentiality of AFA phytocomplex, a new multifunctional cosmetic active ingredient, called AFA-GS, was developed and tested.
Let’s take a look.
What is AFA-GS?
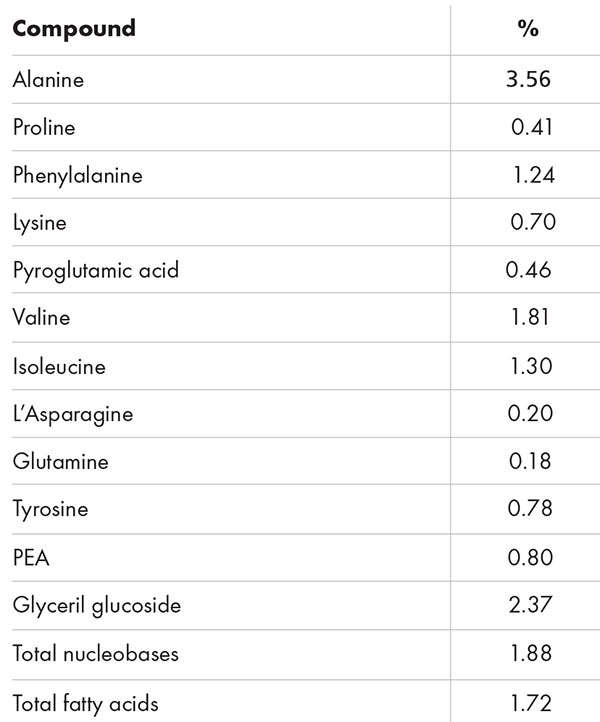
AFA-GS is an innovative cosmetic active ingredient (Ecocert and COSMOS approved) obtained through an internal extraction process from lyophilised Aphanizomenon flos-aquae. The richness of its phytochemical profile was confirmed by GC-MS analysis (table 1).
Several classes of compounds have been identified, such as amino acids (water-binding agents, skin restorers); satured, mono- and polyunsatured fatty acids (with emollient and functional dermopathic activity); and nitrogenous bases (responsible for the improvement of cell tropism and connective matrix deposition, both fibrillary and amorphous).
Moreover, the phytochemical profile is characterised by the presence of phenylethylamine (PEA), a monoamine alkaloid, and of the skin hydration factor and cell-boosting active glyceryl glucoside.
Table 1: GC-MS phytochemical profile of AFA-GS
Anti-radical activity
AFA-GS’ anti-radical activity was evaluated by the conventional DPPH (1,1-diphenyl-2-picryl-hydrazyl) test and the radical inhibition capacity (IC50), expressed as the equivalent content of Trolox (lipophilic analogue of vitamin E) in the product.
AFA-GS showed an IC50 greater than 60% in a concentration of 0.15% (vol/vol), with a Trolox equivalent content of 0.02mgTE/mgAFA-GS.
Anti-inflammatory activity
The AFA-GS anti-inflammatory activity (figure 2) shows a significant reduction of the TNF-α release (ELISA test) on the human monocyte cell line (THP-1), in comparison with the positive control (stimulus with LPS) at all the tested concentrations (0.125-3% vol/vol, based on cytotoxicity data test).
In particular, a high efficacy in the inhibition of TNF-α release, comparable to the negative control (8.08pg/ml), was obtained at concentrations between 2% and 3%.
Figure 2:
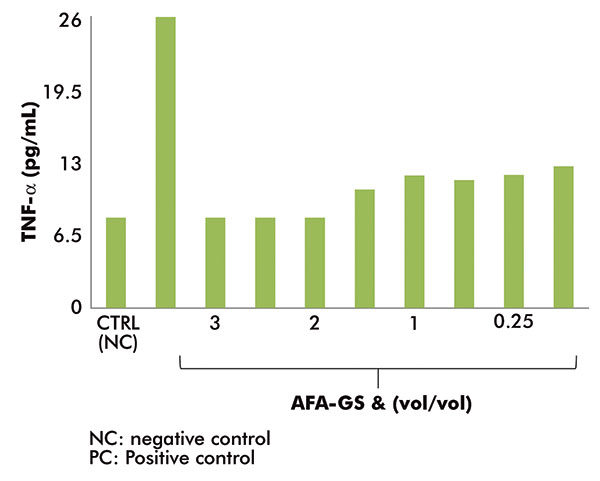
Anti-inflammatory activity of AFA-GS: TNF-α release from LPS-stimulated THP-1 cell line
Anti-pollution activity
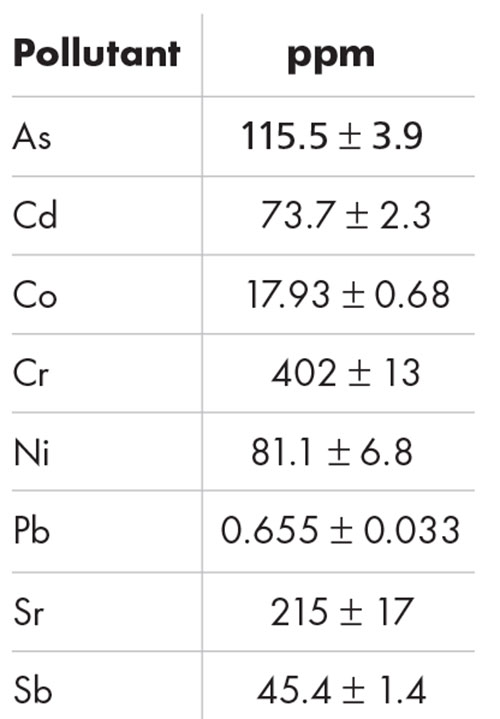
Concern over the impact of atmospheric pollutants on the skin led the formulators to develop different active ingredients to treat related issues, such as skin dryness, loss of firmness, dark spots, inflammation and uneven skin tone.The anti-pollution test was performed on a model of reconstituted human epidermis, in order to evaluate the protective efficacy of AFA-GS on the integrity of the skin barrier exposed to atmospheric pollutants (table 2).
The active (50ml, 3% and 10% vol/vol) was applied on the epidermis units before the stress administration (mixture of air pollutants applied for 24 hours above the stratum corneum).
At the end of the treatment the skin units were subjected to the conventional MTT test (a marker indicative of skin integrity) for the evaluation of cell viability: the release of IL-1α was also evaluated. The treated skin with the only mixture of pollutants was adopted as the positive control (PC), and the untreated epidermis as negative control (NC).
Table 2: Mix of pollutants (heavy metals) applied to the human epidermis
After 24 hours of exposure to atmospheric polluting metals, the PC shows an increase of the release of IL-1α (23.58pg/ml) in respect to the NC (15.78pg/ml).
The application of 3% of AFA-GS did not affected the IL-1α release on the NC (thus demonstrating the non-cytotoxicity of the active), but significantly decreased the inflammatory response on skin treated with pollutants (IL-1α from 23.58 in PC to 18.30pg/ml in PC with 3% AFA-GS).
The results obtained with a 10% concentration of active showed a significant decrease in the release of IL-1α both in NC and in PC (2.17 and 5.68pg/ml, respectively) (figure 3).
Blue light protection
It is known that the violet-blue portion (HEV, high energy visible light, λ=400-500nm) of the visible spectrum induces the formation of ROS, causing skin inflammation and skin ageing.
Figure 3
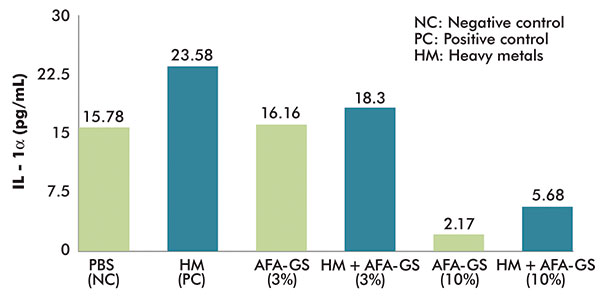
Anti-pollution test: Efficacy of AFA-GS in reducing IL-1α release from reconstituted human epidermis treated with heavy metals
The blue light test was performed in order to evaluate the ROS release by human keratinocytes (Huker) exposed to a Wood lamp (black light) and pretreated with AFA-GS (scalar dilutions from 0.156-2.5mg/ml).
Meanwhile, MTT assay was conducted in order to verify cell viability. The untreated cells represented the negative control (NC).
The obtained results, depicted in figure 4, showed an effective reduction (over 10%) in the ROS formation when keratinocytes were pretreated with AFA-GS at concentrations of 0.156, 0.3125 and 0.625mg/ml.
In summary, AFA-GS, a new active ingredient obtained from alga Klamath, has been shown to significantly reduce the injuries of environmental pollution on skin physiology, acting as a safe anti-radical, anti-inflammatory and anti-ROS agent.
Figure 4
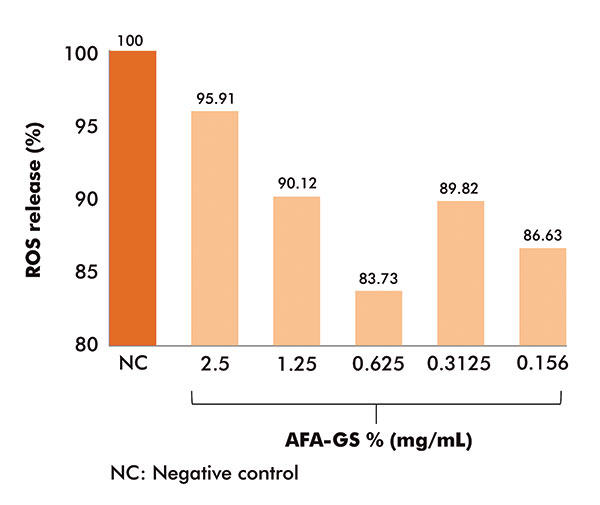
Blue light protection test: Efficacy of AFA-GS in reducing ROS release from human keratinocytes exposed to Wood lamp (4.7mW/cm2)
Furthermore, the presence of the pigment-protein complex C-Phycocyanin confers to the product a protective action against the exogenous and endogenous oxidative damage of mitochondrial DNA, providing an additional tool to counteract skin ageing.
For these reasons, AFA-GS has proven to be an efficient skin detox candidate, acting as an innovative multifunctional cosmetic active ingredient.
References
1. Drakaki et al. 2014. Air pollution and the skin. Frontiers in Environmental Science. 2(11)
2. Munir et al. 2013. Algae: A potent antioxidant source. Sky Journal of Microbiology Research. 1(3)
3. Singh et al. 2010. An efficient method for extraction of C-phycocyanin from Spirulina sp. and its binding affinity to blood cells, nuclei and genomic DNA.International Research Journal of Biotechnology. 1(5)
4. Kay et al. 1991. Microalgae as food and supplement. Critical Reviews in Food Science and Nutrition. 30(6)
5. Pugh et al. 2001. Isolation of Three High Molecular Weight Polysaccharide Preparations with Potent Immunostimulatory Activity from Spirulina platensis, Aphanizomenon flos-aquae and Chlorella pyrenoidosa. Planta Medica. 67(8)
